Welcome to Alberta.
A dynamic ecosystem to conduct clinical trials.
World-class research capabilities, a diverse patient population and an effective ethics framework make Alberta a premier destination to conduct high-quality clinical trials.
About Clinical Trials Alberta

Clinical Trials Alberta is dedicated to establishing Alberta as a premier destination for conducting high-quality clinical trials. Developed as a partnership between academic, government, and research organizations, we work collaboratively to:
- Promote Alberta’s unique strengths to conduct high-quality clinical trials.
- Attract industry R&D investment to the province.
- Provide centralized access for industry to engage with Alberta’s clinical trial assets.
- Enable effective collaboration between industry sponsors and Alberta investigators, sites and service providers.

Our Mission
Our mission is to attract and enable high-quality clinical trials that improve the health of people across the globe.
Our Vision
Our vision is to generate lasting health and economic benefits by solidifying Alberta’s position as a premier clinical trial destination in North America.
Founding Organizations
Clinical Trials Alberta is a collaboration of government, academic institutions, and healthcare delivery organizations working to establish Alberta as a premier destination for conducting high-quality clinical trials.
Clinical Trials Alberta was launched by the partners of the Alberta Clinical Research Consortium (ACRC) – Alberta Innovates, Alberta Health Services, Covenant Health, the College of Physicians & Surgeons of Alberta, the University of Alberta, the University of Calgary, and the Government of Alberta (Alberta Health and Alberta Technology & Innovation).
Our Values
Patient Focus
We understand that our work holds the potential to make a difference in people’s lives. That’s why we go above and beyond to create outcomes that truly matter and work to support the health systems that deliver these outcomes.
Innovation
We leverage Alberta’s entrepreneurial spirit to remove barriers and streamline processes, creating the conditions for breakthrough interventions to reach the people who need them, faster.
Trust
Our commitment to maintaining the highest ethical, methodological and regulatory standards is at the core of everything we do.
Respect and Collaboration
We honour the role that every stakeholder plays in ensuring the success of a clinical trial while recognizing the strength that comes from a diversity of perspectives and backgrounds.
If you're considering where to conduct your next clinical trial—it's time you considered Alberta.
Alberta offers a dynamic clinical trial environment where academic institutes, research organizations and investigators collaborate with industry to pursue the development of new treatments and technologies that improve the health of people across the globe.
That’s why hundreds of industry partners – from startups to multinational organizations – rely on Alberta’s capabilities to conduct groundbreaking clinical trials across a range of therapeutic areas.
Why choose Alberta for your clinical trial?
Our province’s unique advantages enable increased trial efficiencies, reduced operating costs, and faster times to market, all while maintaining the highest quality standards.
Efficiency
Centralized and streamlined provincial processes contribute to efficiency at every stage of the research lifecycle.
Quality
Highly skilled investigators, cutting-edge facilities and an integrated healthcare environment enable high-quality clinical trials.
Incentives
An innovative, business-friendly environment and competitive tax incentives for R&D activities.
News and Stories

IMPACT helps a new medical device find its way to market
The IMPACT Clinical Trial Accelerator Program has a unique mission – to help new ventures and startups in the life science sector commercialize their inventions.
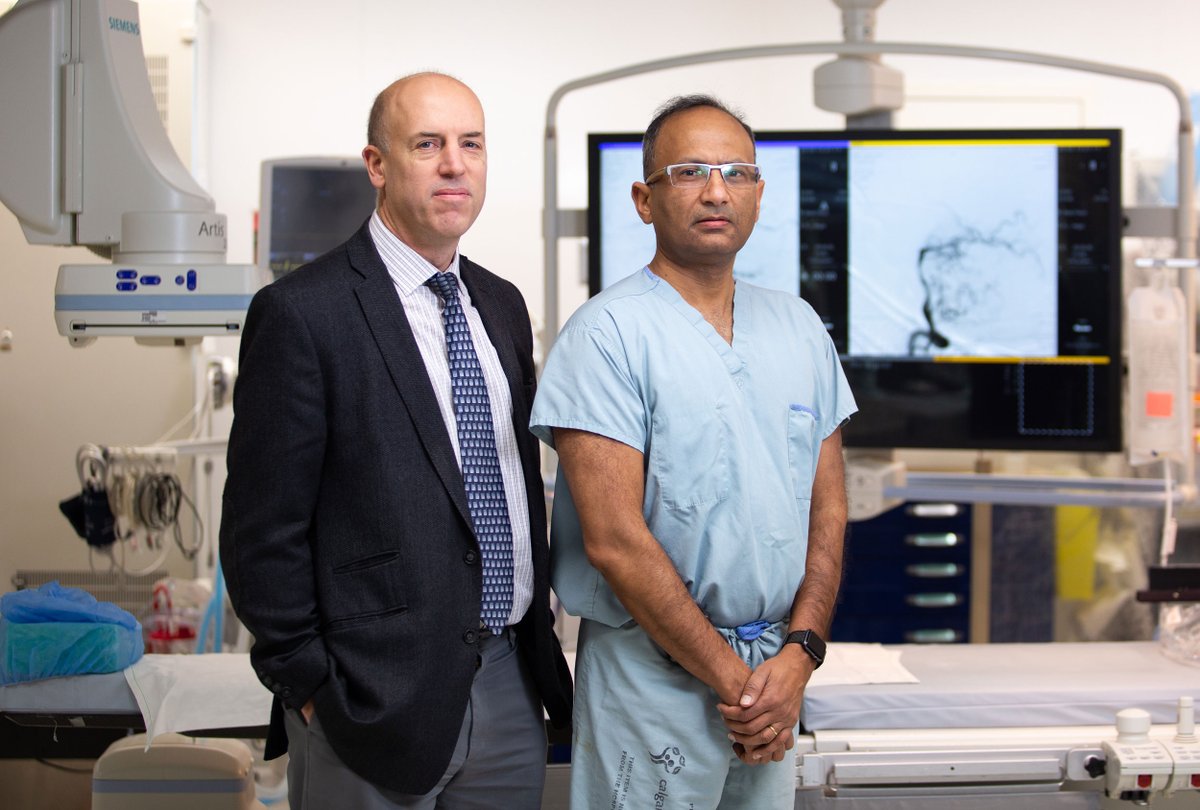
Setting a new standard of care for Ischemic Stroke patients
In 2016, Alberta researchers conducted a groundbreaking multi-site study that changed the global standard of care for Ischemic Stroke.
Creating an informed and supportive participant population
Be The Cure – Alberta’s clinical research public awareness initiative – is creating an informed, primed and supportive participant population.

Alberta’s integrated health system provides tremendous value to provincial COVID research
“Some of the amazing things we accomplished with the CATCO trial would not have been possible to do in many other places,” notes Dr. Tremblay. “It’s a testament to what is possible in our province.”

Introducing the REB Exchange
Get to know Alberta’s innovative technology solution to reduce the administrative burden on researchers and propel new treatments for better patient care outcomes.

Alberta Cell Therapy Manufacturing – Enabling the future of medicine
Regenerative medicine and cell therapies are the future of medicine. They offer the potential to transform the treatment of disease such as cancer and diabetes and the ability to replace damaged tissues and organs, make cartilage for joint repair, and create new therapies to treat autoimmune diseases and organ transplant rejection. The University of Alberta’s Alberta Cell Therapy Manufacturing (ACTM) facility is helping make this future a reality.
Calgary’s Centre for Mobility and Joint Health combines experience with state-of-the-art facilities
Calgary’s Centre for Mobility and Joint Health (MoJo) is one of the province’s most active research imaging facilities. As a key component of the clinical trial infrastructure at the University of Calgary’s Cumming School of Medicine, MoJo is home to state-of-the-art imaging, movement assessment and diagnostic equipment, some of which is only available at a handful of sites worldwide. Complementing this is a highly skilled team of experts and licensed imaging professionals holding extensive research experience.
If you’re seeking an Alberta investigator or site for your clinical trial, we can help.
Clinical Trials Alberta can help you navigate the Alberta research environment and accelerate your trials by:
-
Providing information about conducting clinical trials in Alberta, including regulatory and ethics information.
-
Identifying and connecting you to relevant investigators, research sites, trial networks and service providers.
-
Addressing project-related concerns and challenges.
About this website
This website is meant to provide information to industry about Alberta’s clinical trial capabilities, and offers a central point of access to connect with provincial research assets. Industry organizations are encouraged to learn about the advantages of conducting clinical trials in Alberta and explore opportunities to collaborate with provincial investigators and research sites.
If you’re seeking an Alberta investigator or site for your clinical trial, we can help. Refer to the listing of Alberta Research Assets or reach out to us directly for more tailored information.
This initiative was funded in part by the Ministry of Technology and Innovation.







